By: Ishita Goel
December 7 2023
Old videos passed off as visuals from recent earthquake in Philippines
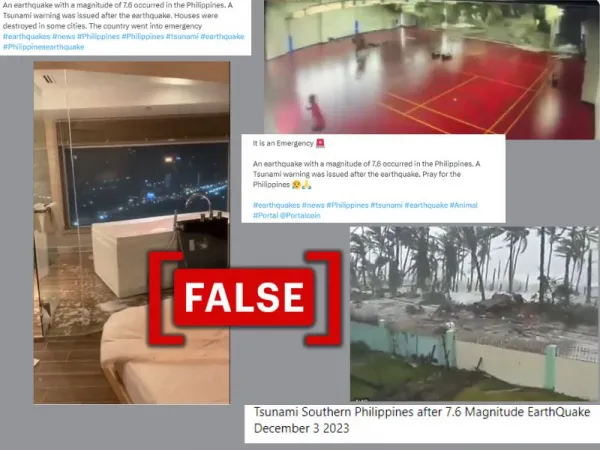
Social media posts claim that videos show the impact of the earthquake and tsunami in the Philippines in 2023. (Source: Facebook/X/Modified by Logically Facts)
The Verdict False
The viral videos of various natural disasters are old, ranging from 2013 to 2022, and are unrelated to the recent earthquake in the Philippines.
A 7.6-magnitude earthquake shook the Province of Surigao del Sur and a large portion of Mindanao in the Philipines on December 2, 2023. At least 600 aftershocks were recorded following the initial quake, with four significant aftershocks of magnitudes exceeding 6.0.
The U.S. Geological Survey measured the first quake at a magnitude of 7.6, triggering tsunami warnings, which were later lifted. On December 4, tsunami waves as high as 0.4m were observed on some of the outlying islands northeast of the Philippines.
What is the claim?
In the aftermath of the earthquake, multiple visuals started circulating on social media, purportedly showing the destruction left behind by the quake. However, some of these visuals included old and unrelated clips that claimed to show damage caused by the recent ‘earthquakes and ensuing tsunamis’ in the Philippines.
Screenshot of claims circulating online, left (first image), upper right (second image), and lower right (third image)(Source: X/Facebook/Modified by Logically Facts)
The first video shows a bathtub with water spilling over, seemingly due to an earthquake. This clip (archived here) was shared with captions claiming that "houses were destroyed in some cities and the country went into emergency" because of the earthquake in the Philippines. A user shared the clip on Facebook (archived here) with the caption: "The Philippines was shaken near Bislig City, Surigao del Sur, Caraga, by an earthquake of magnitude 7.5. Power can be seen by water spilling out of the tub." Other archived versions of such posts can be accessed here and here.
The second video captures a sports center's ceiling collapsing while people are playing, linked to the Philippine earthquake with dramatic captions. The video was shared with this caption on X (archived here): "Video of the damage caused by the #earthquake that has hit in Philipines." Other such captions read, "It is an Emergency. An earthquake with a magnitude of 7.6 occurred in the Philippines. A Tsunami warning was issued after the Earthquake. Pray for the Philippines". Archived versions of these posts can be accessed here and here.
The third video depicts trees swaying in heavy winds, houses flooded by high waves, and debris being swept away. This was shared with claims showing the aftermath of a tsunami in Southern Philippines following the 7.6 magnitude earthquake (archived here and here).
However, our investigation revealed that all these videos are old and unrelated to the recent earthquake in the Philippines.
Video of bathtub
A reverse image search traced the first video back to 2021. The Weather Channel posted it on February 23, 2021, with the caption: “Watch Water Leap From Tub During Japan Earthquake.” The location was identified as Chiba City, Japan. Two separate YouTube channels also uploaded extended versions of the clip, attributing it to an earthquake in Japan. A 7.3-magnitude quake was indeed reported in Fukushima, Japan, around this time, affecting power and water supplies.
The video was published by the Weather Channel and YouTube in 2021. (Source: Website/YouTube/Modified by Logically Facts)
Video of sports center
The second video, featuring the sports center roof collapse, is from 2022. Sky News and CNN covered this incident, reporting it as part of a 6.9 magnitude earthquake in Taiwan. Geolocation confirmed the site as Taoyuan City Bade Civil Sports Center in Taiwan. Reuters also reported on this earthquake, noting its widespread impact across Taiwan. A photo posted in November 2021 by Sky News matches a frame from the viral video.
The video was published by the Sky News and CNN in 2022. (Source: Website//YouTubeModified by Logically Facts)
Video showing flooding and heavy winds
The third video dates back to at least 2013. We found the video posted by multiple YouTube users over the last few years, the oldest being from November 2013. It was initially captioned as footage of the aftermath of Typhoon Haiyan (Yolanda) in Leyte, Philippines.
Another YouTuber shared the video in August 2018 and wrote, "Typhoon Haiyan, known as Super Typhoon Yolanda in the Philippines, was one of the strongest tropical cyclones ever recorded."
A Philippine-based television and radio network TV5 shared a screenshot of the video on Facebook on December 5, 2014, and wrote in Filipino, "THIS IS THE EFFECT OF A 'STORM SURGE' This video shows the impact of the storm surge in Eastern Samar and Leyte in 2013."
The video was published on YouTube and TV5 in 2013. (Source: YouTube/Website/Modified by Logically Facts)
Typhoon Haiyan, known as Super Typhoon Yolanda, made landfall in the Philippines between November 3 and 11, 2013. CNN and BBC covered Typhoon Haiyan, describing it as one of the strongest storms recorded, with significant damage and loss of life.
Logically Facts could not independently verify the place of origin of some of these videos. However, they have existed on the internet well before the recent earthquake in the Philippines.
The verdict
Old videos from various natural disasters, ranging from 2013 to 2022, have been falsely shared as visuals from the recent Philippines earthquake and its aftermath. These findings lead us to label the claim as false.


