By: Emincan Yüksel
November 24 2023
No, this video does not show a priest in a church protesting against Israel
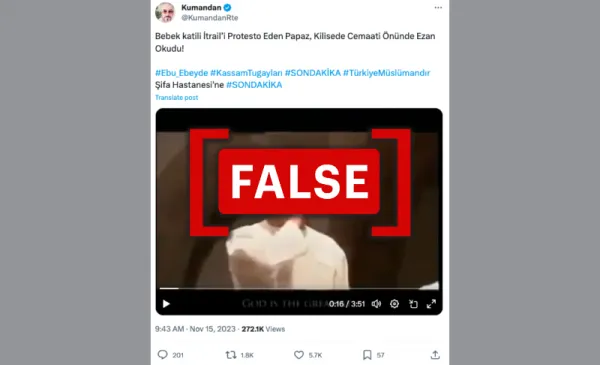
Post featuring claims that the call to prayer video is in protest against Israel (Source: YouTube/LF)
The Verdict False
This video from 2009 shows Algerian actor and muezzin Ben Youcef making a call to prayer in a church and is not related to the Israel-Hamas war.
Context
On November 15, 2023, a social media account in Türkiye shared a post on X with a video claiming to show a priest making a call to prayer to protest against Israel. The post and video received over 270,000 views and 5,700 likes in one week. Multiple accounts on X and Facebook also shared the video. The same video has circulated on social media since 2017, titled, "Priest is calling the Azan in church."
In fact
A detailed search on YouTube shows that the video was first published on November 2, 2009, with the caption "Ben Youcef: Interfaith Service For Peace (Full Version)" and has no connection to the Israel-Hamas war. It was filmed at All Saints Church in California, U.S., and belongs to the Prayers For Peace organization, an interfaith community of Christians, Jews, and Muslims.
(Source: YouTube, Ben Youcef: Interfaith Service for Peace (Full Version))
The man in the video is Algerian actor Ben Youcef, a muezzin, not a priest. His official site describes his membership in the Interfaith community of Jews, Christians, and Muslims in LA. The "Call to Prayer" video has gone viral on YouTube.
Ed Bacon, who speaks at the beginning of the video, is the former priest (reverend) of Pasadena's All Saints Church. In his speech, he said the following: "We who are here gathered in California this day extend our love, our peace, our goodwill to all sisters and brothers of ours in the human family who will view this documentary of this moment in which all of us were gathered with one desire, one commitment; to make peace in our earth and to bring an end to religion blessing war. Thank you for being here, and to all who will view this. We extend you our deepest wishes for peace. Salaam. Shalom."
Reverend Ed Bacon makes a speech before Ben Youcef makes a call to prayer. (Source: YouTube/Screenshot)
Bacon's speech makes it clear that the call to prayer in the church is not a protest against Israel but a call for world peace.
The verdict
This video does not show a priest making a call to prayer inside a church in protest against Israel. The video was uploaded to YouTube in 2009 and is unrelated to the Israel-Hamas conflict or protest against Israel. The call to prayer featured in the video is not called by a priest but by the Algerian actor and muezzin Ben Youcef. Therefore, we have marked this claim as false.


